The market for ready-to-use (RTU) syringes is growing rapidly. In fact, experts anticipate an annual growth rate of about eight percent over the next ten years.
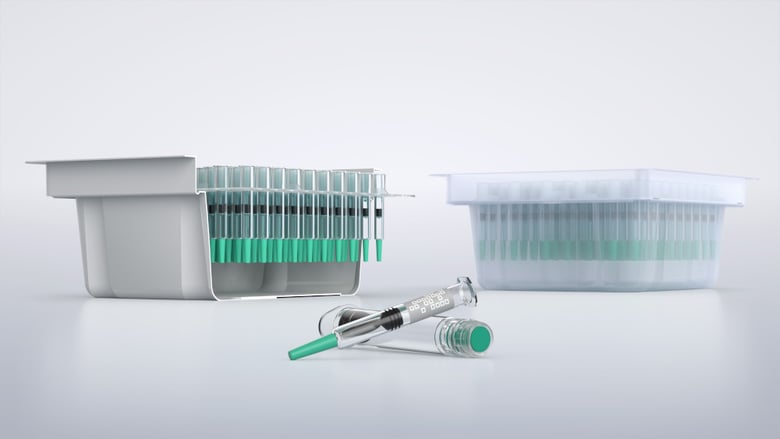
The market for ready-to-use (RTU) syringes is growing rapidly. In fact, experts anticipate an annual growth rate of about eight percent over the next ten years. In response to this demand, Syntegon has expanded its inspection portfolio with the new AIM DIRect to include integrated de-nesting and re-nesting technologies for nested syringes into its inspection lines.

Benefits and challenges of RTU syringe nests
RTU syringes in a nest offer secure handling and optimal protection. Syringe nests help organize syringes efficiently, ensuring they are stored compactly and safely. They provide crucial protection during both storage and transportation by keeping syringes in a stable, upright position, which minimizes the risk of breakage.
Since inspection is a crucial step in ensuring the quality of pharmaceuticals, manufacturers using RTU syringes in their production need reliable solutions for the automated handling of syringes and their nests, such as de-nesters and re-nesters. However, there are challenges associated with syringe nests during the inspection process. Improper handling can lead to air bubbles, which may increase the risk of false rejects. Additionally, only fully filled syringe nests should be delivered after inspection, ensuring each nest contains the correct number of syringes.
Syntegon Newsletter
Get inspired with regular updates on technologies and services, case studies, webinar offers and events.

See our new AIM DIRect in action
Interested in a live demonstration of our de-nester and re-nester solution? Visit our shop floor to experience it firsthand and discover how our other inspection technologies can help you meet the latest safety standards. The whole team is looking forward to welcoming you here are our Center of Competence for Inspection in Japan.
Get in touch with our global product manager Leoni Ransmann today to arrange a visit!



