By end of December 2019, the Russian government published regulatory guidelines regarding Track & Trace for cosmetic products. Aim is to inform and protect customers from counterfeits. Since the deadlines are quite close, we have set up all information regarding necessary activities and the ways of implementing in time.
Russian Governmental Resolution No. 1957, dated December 31st 2019, forms the basis for this challenging situation. Jörg Willburger, Product Manager for Track & Trace, sees synergies to other industries: “Processes and infrastructure for cosmetic industry will not differ too much from the pharmaceutical solution. A similar system to proven CRPT/MDLP system will be applied, which is well known in the pharma industry.”
Implementation deadlines
Currently only Perfumes and Eau de Toilettes (defined by customs tariff number) are affected, but it is still uncertain if other cosmetic products will be included. Like in pharma, the regulatory guidelines intend to inform and protect consumers by increasing transparency of supply chains. Following timeline points out the tight schedule till implementation deadlines:
- Already till March 31, 2020 all trade participants of Perfumes and Eau de Toilettes have to be registered in the MDLP system (alternatively max. 7 days after the start of trading with the corresponding products).
- October 1, 2020: All consumer packaging of Perfume / Eau de Toilette produced or imported into Russia must be provided with a data matrix code and other notifications.
- All information must be transmitted to the CRPT/MDLP system. However, the sale of unmarked products is only permitted until September 30, 2021 if they were produced in Russia or imported to Russia before October 1st, 2020.
Technical details and marking requirements
Let´s have a quick view on technical details for MDLP/CRPT system and marking requirements:
Marking (serialization) of consumer packaging (directly onto the carton/bottle or by a label) must be done by a 2D data matrix code (75 symbols), according to ISO 16022-2008. The 2D data matrix code includes 4 data groups:
- AI01 (14 symbols for Identnumber)
- AI21 (13 symbols for Serialnumber)
- AI91 (4 symbols as Check-Key)
- AI92 (44 symbols as “cryptocode”)
Marking (aggregation) on bundle and/or case/pallet level with information about each serial number has to be done by SSCC code according ISO 15394-2013. These packaging levels must be also marked with customs tariff number according to the rules of the Eurasian customs unions in a 1D barcode.
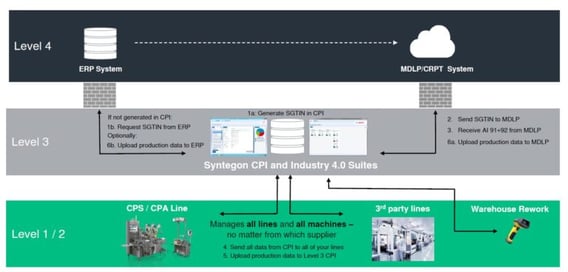
The process to get the required crypto data is described in the chart above. Customers are free to choose how to generate the serial numbers: either by CPI (Syntegon´s software solution for data handling) or in their ERP system. After the serial numbers are generated, they are transferred to CRPT/MDLP system and “as an answer”, the crypto data is received. As soon as production is completed, all relevant data are uploaded to CRPT/MDLP system. Communication with MDLP can be done via CPI as shown above or – if requested and if an interface is available – between the ERP and CRPT/MDLP system. Due to flexibility and modularity, the Syntegon software CPI is able to meet all customer specific architectures.
“Syntegon not only offers the mechanical solution to fulfill the latest regulations released by Russian government. Also software solutions for line and site management, as direct interface to CRPT/MDLP system and proven interfaces to 3rd party systems like SAP, TraceLink or 1C can be provided. Additionally, we offer MES and Industry 4.0 solutions. Don’t hesitate to contact our expert team” states Ralf Weihbrecht, Sales Director for Secondary Packaging, Inspection and Track and Trace, “we support you in implementing necessary processes right in time.”